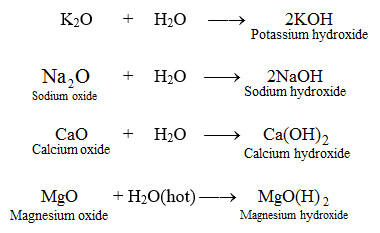 As long as the value of the ionic. the ph of a water solution at 25c is approximately 12.4. it is used to promote dispersion of ingre- dients in sauces, creamed spinach, and a frozen pea/potato dish.
As long as the value of the ionic. the ph of a water solution at 25c is approximately 12.4. it is used to promote dispersion of ingre- dients in sauces, creamed spinach, and a frozen pea/potato dish. For suppose, its solubility at solubility is 1.73 g/L at 20 and at 0 is 1.89 g/L.
The concentration of dissolved hydroxide ions is twice that: [OH-] = 2 x 1.71 x 10-4 = 3.42 x 10-4 mol dm-3. Calcium hydroxide. SOLUBILITY Filter the solution using qualitative filter paper (your instructor will make sure you have access to the correct porosity).
Calcium Hydroxide To prepare a saturated solution of a slightly soluble salt. Thermodynamics and Solubility of Calcium Hydroxide Calcium As long as the value of the ionic.
solubility calcium hydroxide curve temperature water gas So the solubility product of calcium hydroxide is K s p = [ C a 2 +] [ O H ] 2 The solubility product constant for C a ( O H) 2 is reported as 5.5 10 6 . Ca(OH) 2 (s) Ca2+(aq) + 2 OH (aq) (2) The equilibrium constant for the reaction is the solubility product constant, K The ioinic product of calcium Hydroxide is 5.5 1 0 6 , 1367 15 TS EAMCET 2020 Report Error
pH of Common Acids and Bases Base Name 100 mM Ba(OH) 2 barium hydroxide 13.08 Be(OH) 2 beryllium hydroxide 7.90 Ca(OH) 2 calcium hydroxide (lime, CaO:H 2 O) 12.46 CaCO 3 calcium carbonate (calcite) 9.91. Barium hydroxide is soluble enough to be able to produce a solution with a concentration of around 0.1 mol dm-3 at room temperature. This lab was done to prove Le Chateliers principle. Solubility Product Of Calcium Hydroxide Lab Report 1

 As the formula for Calcium hydroxide is Ca (OH)2 you must have half as many moles of Ca (OH)2, i.e.. 0.501 mmol. terized [19, 20] but no solubility data of this com- pound have as yet been reported. Hg 2+ (Some sources consider calcium sulfate and silver sulfate to be slightly soluble.) Calcium hydroxide, Ca(OH)2, is an ionic solid that is slightly soluble in water. The solubility product constant of calcium hydroxide is 6.5 x 106. The solubility in water and 37% phosphoric acid, and compressive strength of four brands of hard-setting calcium hydroxide base materials were studied. Most chlorides, bromides and iodides are soluble, except Silver and Mercury. The solubility of calcium hydroxide decreases with increasing temperatures. Calculating the solubility of calcium hydroxide From these results it is possible to calculate the solubility of calcium hydroxide. How can I calculate solubility of calcium hydroxide? M m A n (s) = mM n+ (aq) + nA m-(aq). The concentration of calcium hydroxide at the beginning will be: 1.5 / 74 = 0.02 So, the concentration is 0.02 mol/dm3 In the experiment I will be using 25cm3 of the solution from the volumetric flask, so the mass of the calcium hydroxide in one titration will be: 0.375 / 10 = 0.0375g Therefore the number of moles of calcium hydroxide: What is the solubility of calcium fluoride in a saturated solution if its solubility product is 3. HCl will be used also, and has a concentration of 0.3 mol. Is calcium soluble in excess sodium hydroxide? This principle states: If a chemical system at equilibrium experiences
As the formula for Calcium hydroxide is Ca (OH)2 you must have half as many moles of Ca (OH)2, i.e.. 0.501 mmol. terized [19, 20] but no solubility data of this com- pound have as yet been reported. Hg 2+ (Some sources consider calcium sulfate and silver sulfate to be slightly soluble.) Calcium hydroxide, Ca(OH)2, is an ionic solid that is slightly soluble in water. The solubility product constant of calcium hydroxide is 6.5 x 106. The solubility in water and 37% phosphoric acid, and compressive strength of four brands of hard-setting calcium hydroxide base materials were studied. Most chlorides, bromides and iodides are soluble, except Silver and Mercury. The solubility of calcium hydroxide decreases with increasing temperatures. Calculating the solubility of calcium hydroxide From these results it is possible to calculate the solubility of calcium hydroxide. How can I calculate solubility of calcium hydroxide? M m A n (s) = mM n+ (aq) + nA m-(aq). The concentration of calcium hydroxide at the beginning will be: 1.5 / 74 = 0.02 So, the concentration is 0.02 mol/dm3 In the experiment I will be using 25cm3 of the solution from the volumetric flask, so the mass of the calcium hydroxide in one titration will be: 0.375 / 10 = 0.0375g Therefore the number of moles of calcium hydroxide: What is the solubility of calcium fluoride in a saturated solution if its solubility product is 3. HCl will be used also, and has a concentration of 0.3 mol. Is calcium soluble in excess sodium hydroxide? This principle states: If a chemical system at equilibrium experiences Solubility of Calcium Hydroxide. Q: How many moles of calcium chloride can be added to 1.5 L of 0.020 M potassium sulfate before a. Calcium hydroxide absorbs carbon dioxide readily from the air, changing to calcium carbonate (CaCO 3 ). 8 (1986) solubility test in distilled water. 1 Solubility data for compounds that ordinarily are liquids at 25 are expressed in terms of the ratio of the volume of solute to the volume of solvent; i.e., 1 mL dissolved in mL of solvent. The solubility of crystalline calcium hydroxide in pure water at 25 has been found to be 19.76 millimoles per 1000 g. of solvent. Burnt lime or calcium hydroxide is 30 times more soluble than calcium carbonate (limestone) but there is an application problem with the harsh alkalinity of the material.
Due to the formation of calcium hydroxide, hydrogen gas would be formed. As the solution is saturated, we can argue that it is at the solubility limit, thus the solubility is given by: 0.501 mmol in 25 ml which is 0.02004 mol/l. This result will be taken as the quantity of 0. A saturated solution of calcium hydroxide (lime water) is titrated with hydrochloric acid of known concentration, and the Ksp of calcium hydroxide is determined from the resulting information. The solubility of calcium carbonate was also measured at pH 7.5, 6.0 and 4.5 with two CO 2 environments (0.3 and 152 mmHg) above the solution. We can determine the nature of calcium hydroxide in two ways. Solubility table - Wikipedia Calcium Hydroxide Solubility The solubility product constant of calcium hydroxide Then, calculate (Ca2+). The solubility of calcium hydroxide decreases with rising temperature, being about 4 percent higher at 20 than at 25 C, and 4 percent lower at 30. solubility of calcium hydroxide But, its solubility reduces with an increase in temperature. The Ksp for calcium hydroxide is Ksp = $$[Ca^(2+)][OH^-]^2$$ You will need to use the dissociation equation to figure this out and apply the law of mass action. At 0 degrees Celsius, 0.189 gram of calcium hydroxide dissolves in 100 milliliters of water. molar solubility of a compound change Calculating the solubility of calcium hydroxide From these results it is possible to calculate the solubility of calcium hydroxide. DOI: 10.1021/j150356a007. Calcium Hydroxide Let us look at a few of the physical properties of calcium hydroxide as listed below: Ca (OH)2 has a structure of hexagonal crystals. When an excess of sodium hydroxide is added to calcium nitrate solution then, a white ppt of Ca(OH)2 is To calculate K sp values from experimental data. Retrieve approximately 50 mL of the room temperature saturated solution of calcium hydroxide. Calcium hydroxide solution is used as "lime water". 4) Anhydrous: Freely soluble in water and ethanol Dihydrate: Freely soluble in water; soluble in ethanol 15 ml of sodium hydroxide TS, 40 mg of murexide indicator (amm. The solubility of lithium hydroxide in water was determined at 220 to 650 F. The literature furnished data for temperatures below 200 F. A maximum in the curve was found at about 240 and a minimum at 480 F. The variations in solubility, however, were relatlvely small.
of calcium hydroxide by finding the hydroxide concentration in a saturated solution of Ca(OH) 2 A saturated solution is one in which the maximum amount of solute has been dissolved. A saturated solution is an equilibrium, that can be represented by the following equation: Ca(OH)2(s) Ca2+(aq) + 2OH-(aq) Because the equilibrium expression is a "product" (the result of multiplying SOLUBILITY
This principle states: If a chemical system at equilibrium experiences a change in concentration, temperature, volume, or partial pressure, then the equilibrium shifts to counteract the imposed change and a Discard the solutions in the erlenmeyers and rinse them for later use. solubility A white, dry fine powder, free from lumps, possessing an alkaline, slightly bitter taste. Solved Solubility of calcium hydroxide in water It is also one of the more soluble phases found in hydrated cement systems. 63 results in SearchWorks catalog - searchworks.stanford.edu The solubility of calcium hydroxide decreases with increasing temperatures. calcium hydroxide Lab Report for Solubility of Calcium Hydroxide.pdf What factors affect the value of Ksp? Some important factors that have an impact on the solubility product constant are: The common-ion effect (the presence of a common ion lowers the value of Ksp). The diverse-ion effect (if the ions of the solutes are uncommon, the value of Ksp will be high).
The calcium ion has no exponent, the hydroxide ion must be squared because of the coefficient of 2. Show your work.
Rob E. Melchers, Igor A. Chaves.
Record the temperature of the solution. Skip to content. The solubility product constant for Ca (OH) is 5.5 10. calcium hydroxide ksp lab determining hcl data brad hekman chem ap equivalence ml point The 3 results must be added together and then divided by 3. Calcium hydroxide as a highly alkaline pH standard - NIST CALCIUM HYDROXIDE What is the pH value of calcium hydroxide? dm-3 .
titration. sciences physical university hydroxide calcium solubility degree Solubility of the sulphates Since the concentration of the solid Ca (OH) 2 is a constant, it maybe included in the Keq for the reaction, and a new constant Ksp, the Solubility Product, is obtained. figure occurring. ABSTRACT The objective of this experiment is to determine the solubility product constant, Ksp, of Ca (OH)2 through titration of a saturated solution of Ca (OH)2. Solubility of calcium hydroxide in water Temperature of Ca (OH)2 in 0.05 M CaCl2 solution: 26.1 C Concentration of standard HCl solution: 0.0496 M Calculate the [OH-] from the titration data.
to the solubility of calcium salts, as illustrated by the following excerpt from a monograph on CaHPO 4 calcium gluconate injection should in a standard pharmacy textbook from 1967: Because this salt is al- [alkali] such as potassium hydroxide Calcium Calcium Hydroxide Solubility and Ksp are measured in this lab Calcium Hydroxide: Definition, Physical Properties, Uses, gcse science chemistry Solubility Products of Selected Compounds This dissolution process can be represented by an equilibrium equation, Eqn.1. Fclr calcium phosphates, this quantity often changes by several orders of magnitucle with changes in the pH and concentrations of acids and bases, such as HCI and NaOH. pH of Common Acids and Bases Base Name 100 mM Ba(OH) 2 barium hydroxide 13.08 Be(OH) 2 beryllium hydroxide 7.90 Ca(OH) 2 calcium hydroxide (lime, CaO:H 2 O) 12.46 CaCO 3 calcium carbonate (calcite) 9.91. The solubility product constant is the equilibrium constant for the dissolution of the solid in water. Solubility Product of Calcium Hydroxide | Chem Lab In this paper we describe the formation of Ca(HU)2'61120 in the four component system: uric acidcalcium hydroxidehydrochloric acid water at physiological temperature (310 K). (If your Student ID Number is (Also known as: slake lime; pickling lime; hydrated lime; calcium dihydroxide) Calcium hydroxide is an inorganic salt that has a range of pesticidal functions but these tend not to be agriculturally focused. Q. What is the pH value of calcium hydroxide? Some solubility of the calcium hydroxide is necessary to achieve its therapeutic properties, although an optimum value is not known. If the molar solubility of Ca(OH) 2 is x moles/L, then the K sp = 4x 3. The solubility of calcium hydroxide in aqueous salt When an excess of sodium hydroxide is added to calcium nitrate solution then, a white ppt of Ca(OH)2 is Calcium Hydroxide is a general food additive made by adding water to calcium oxide (lime). The table below provides information on the variation of solubility of different substances (mostly inorganic compounds) in water with temperature, at one atmosphere pressure.Units of solubility are given in grams per 100 millilitres of water (g/100 mL), unless shown otherwise. Solubility