Leaching of pesticides through normal-tillage and low-tillage soil--a lysimeter study. However, NIST makes no warranties to that effect, and NIST  Evolution over time of the agricultural pollution of waters in an area of Salamanca and Zamora (Spain). suberin monomer interactions pesticides conformations isoproturon adducts Therefore, Hirshfeld partitioning scheme was used to derive the atomic charges at the MP2/6-311++G(2d,2p) level. Amylase induction during seed germination in isoproturon susceptible and resistant biotypes of Phalaris minor Retz. Often supplied as a soluble concentrate that is mixed with water and used as a spray; New low dose formulations being considered for UK registration. An agrochemical is a substance that is used in agriculture or horticulture. Pesticides in rainwater in Flanders, Belgium: results from the monitoring program 1997-2001. Mineralization of soil-aged isoproturon and isoproturon metabolites by Sphingomonas sp. Seasonal changes in the sensitivity of river microalgae to atrazine and isoproturon along a contamination gradient. Pesticide residues on the external surfaces of field-crop sprayers: environmental impact.
Evolution over time of the agricultural pollution of waters in an area of Salamanca and Zamora (Spain). suberin monomer interactions pesticides conformations isoproturon adducts Therefore, Hirshfeld partitioning scheme was used to derive the atomic charges at the MP2/6-311++G(2d,2p) level. Amylase induction during seed germination in isoproturon susceptible and resistant biotypes of Phalaris minor Retz. Often supplied as a soluble concentrate that is mixed with water and used as a spray; New low dose formulations being considered for UK registration. An agrochemical is a substance that is used in agriculture or horticulture. Pesticides in rainwater in Flanders, Belgium: results from the monitoring program 1997-2001. Mineralization of soil-aged isoproturon and isoproturon metabolites by Sphingomonas sp. Seasonal changes in the sensitivity of river microalgae to atrazine and isoproturon along a contamination gradient. Pesticide residues on the external surfaces of field-crop sprayers: environmental impact.
CopyCopied, CSID:33695, http://www.chemspider.com/Chemical-Structure.33695.html (accessed 20:42, Jul 29, 2022)
Effects of the pesticides captan, deltamethrin, isoproturon, and pirimicarb on the microbial community of a freshwater sediment. weeds in cereals, its use within the EU has been banned after September 2017 on the grounds of potential groundwater contamination and risks to aquatic life; there have also been concerns about its en
Lewis, K.A., Tzilivakis, J., Warner, D. and Green, A. Characterization of aqueous and solid inclusion complexes of diuron and isoproturon with beta-cyclodextrin. If the file has been modified from its original state, some details such as the timestamp may not fully reflect those of the original file. In other study, the fragmentation pathways of the anionic forms of diuron were investigated employing the Density Functional Theory (DFT) [38] while the structure and vibrational spectrum of chlorotoluron and isoproturon were analyzed by Q. Wang and H. L. Wang [39] and Vrielynck et al. On the other hand, the atom 7C is susceptible to either nucleophilic or free radical substitutions. Metallothionein induction in the aquatic oligochaete Tubifex tubifex exposed to the herbicide isoproturon. Dihedral angle 2C-4C-10N-12C = 17.53, 10N-12C-13N-21O = 18.34. However, it has been reported that AOPs may produce harmful intermediates [3032], while microbial and fungal degradation methods are unable to degrade completely these PHUs [29, 33]. The Fukui function values suggest that electrophilic attacks on chlorotoluron, linuron, and diuron herbicides would cause the elimination of the urea moiety while electrophilic, nucleophilic, or free radical attacks on isoproturon would eliminate the isopropyl moiety in aqueous conditions. All rights reserved. isoproturon D'Archivio, M. Fanelli, and S. Santucci, Photocatalytic degradation of linuron in aqueous suspensions of TiO, H. Katsumata, M. Sada, Y. Nakaoka, S. Kaneco, T. Suzuki, and K. Ohta, Photocatalytic degradation of diuron in aqueous solution by platinized TiO, A. Amorisco, I. Losito, T. Carbonara, F. Palmisano, and P. G. Zambonin, Photocatalytic degradation of phenyl-urea herbicides chlortoluron and chloroxuron: characterization of the by-products by liquid chromatography coupled to electrospray ionization tandem mass spectrometry,, S. Aquino and A. Rodrigues, Electrochemical oxidation of herbicides, in, S. Nlieu, F. Perreau, F. Bonnemoy et al., Sunlight nitrate-induced photodegradation of chlorotoluron: evidence of the process in aquatic mesocosms,, Y. C. Lu, S. Zhang, S. S. Miao et al., Enhanced degradation of herbicide isoproturon in wheat rhizosphere by salicylic acid,, P. Miriti, G. Magoma, H. I. Boga, and A. Nyende, Isolation and characterization of linuron degrading bacteria from soils under horticultural production in Kenya,, J. Safi, Y. Awad, and Y. El-Nahhal, Bioremediation of diuron in soil environment: influence of cyanobacterial mat,, G. A. Turnbull, M. Ousley, A. Walker, E. Shaw, and J.  Here, it is interesting to highlight that the values of the Fukui Function found for diuron (see Figure 7) and linuron (see Figure 8) indicate that the more reactive sites are located on 4C, 7C, and 7C. strain SRS2. Data Program, but require an annual fee to access. on behalf of the United States of America. In soils, it has been observed that these herbicides may be degraded through demethylation at the urea nitrogen by mean enzymatic and microbial activity [34]. P. A. Johnson, L. J. P. Bartolotti, W. Ayers, T. Fievez, and P. Geerlings, S.-B. National Center for Advancing Translational Sciences (NCATS), N,N-DIMETHYL-N'-(4-(1-METHYLETHYL)PHENYL)UREA, U.S. Department of Health & Human Services. isoproturon The bond distances and angles obtained at the MP2/6-311++G(2d,2p) levels are comparable with those reported in the literature [68, 69]. Pesticide degradation in a 'biobed' composting substrate. [40], respectively. Rapid mineralisation of the herbicide isoproturon in soil from a previously treated Danish agricultural field.
Here, it is interesting to highlight that the values of the Fukui Function found for diuron (see Figure 7) and linuron (see Figure 8) indicate that the more reactive sites are located on 4C, 7C, and 7C. strain SRS2. Data Program, but require an annual fee to access. on behalf of the United States of America. In soils, it has been observed that these herbicides may be degraded through demethylation at the urea nitrogen by mean enzymatic and microbial activity [34]. P. A. Johnson, L. J. P. Bartolotti, W. Ayers, T. Fievez, and P. Geerlings, S.-B. National Center for Advancing Translational Sciences (NCATS), N,N-DIMETHYL-N'-(4-(1-METHYLETHYL)PHENYL)UREA, U.S. Department of Health & Human Services. isoproturon The bond distances and angles obtained at the MP2/6-311++G(2d,2p) levels are comparable with those reported in the literature [68, 69]. Pesticide degradation in a 'biobed' composting substrate. [40], respectively. Rapid mineralisation of the herbicide isoproturon in soil from a previously treated Danish agricultural field.
Luis Humberto Mendoza-Huizar gratefully acknowledges financial support from CONACYT (Project INFR-2014-227999) and the Universidad Autnoma del Estado de Hidalgo through Projects PIFI 2008-13M8U0017T-04-01 y PIFI-2009-13MSU0017T-04-01.
M. J. Frisch, G. W. Trucks, H. B. Schlegel et al., S. Gunasekaran, R. K. Natarajan, D. Syamala, and R. Rathikha, Normal Coordinate ananlysis of urea meta nitro benzoic acid crystal,, T. Vroumsia, R. Steiman, F. Seigle-Murandi, J.-L. Benoit-Guyod, and A. Khadrani, Biodegradation of three substituted phenylurea herbicides (chlortoluron, diuron, and isoproturon) by soil fungi.
A member of the class of phenylureas that is 1,1-dimethylurea substituted by a p-cumenyl group at position 3. Dihedral angle 12C-11C-6C-3C = 0.2, 2C-4C-20N-22C = 22.03. at the th atomic site in the neutral (), anionic (), or cationic () chemical species. Standard Reference Data Act. Evaluating non-equilibrum herbicides transport in undisturbed grassed buffer strips soil columns. Effect of soil composition and dissolved organic matter on pesticide sorption. molecule skeletal herbicide isoproturon glucoside  Additional to the global reactivity parameters, it has been possible to define local reactivity parameters [52, 53], such as the Fukui Function () [54, 55]. isoproturon monodes ipu mineralization metabolites Study of solubility of isoproturon by its complexation with beta-cyclodextrin. Also, the structures optimized at the B3LYP/6-311++G(2d,2p) level were further reoptimized at the MP2/6-311++G(2d,2p) level in the aqueous phase; see Figures 1, 2, 3, and 4.
Additional to the global reactivity parameters, it has been possible to define local reactivity parameters [52, 53], such as the Fukui Function () [54, 55]. isoproturon monodes ipu mineralization metabolites Study of solubility of isoproturon by its complexation with beta-cyclodextrin. Also, the structures optimized at the B3LYP/6-311++G(2d,2p) level were further reoptimized at the MP2/6-311++G(2d,2p) level in the aqueous phase; see Figures 1, 2, 3, and 4.  Dihedral angle 2C-4C-10N-12C = 17.53, 10N-12C-13N-21O = 18.34. Evaluation and characterization of a commercial immunosorbent cartridge for the solid-phase extraction of phenylureas from aqueous matrices. A. EU Annex III PIC DGD) (, R = Peer reviewed scientific publications, L = Pesticide manuals and hard copy reference books / other sources, A = Chromosome aberration (EFSA database), E = Unspecified genotoxicity type (miscellaneous data source), (Also known as: IPU; ipuron; CGA 18731; AE F016410), A herbicide for use, typically, in cereals to control annual grasses and many broad-leaved weeds, Cereals including wheat, barley, rye; Rice-wheat cropping systems, EC Regulation 1107/2009 (repealing 91/414), Approved for use () under EC 1107/2009 in the following EU Member States, International Chemical Identifier key (InChIKey), International Chemical Identifier (InChI), InChI=1S/C12H18N2O/c1-9(2)10-5-7-11(8-6-10)13-12(15)14(3)4/h5-9H,1-4H3,(H,13,15), Cambridge Crystallographic Data Centre diagrams. Selective, systemic absorbed by roots and leaves. 1SD, UK +44 (0)1223 49 44 44, Copyright EMBL-EBI 2018 | EBI is an outstation of the European Molecular Optical whole-cell biosensor using Chlorella vulgaris designed for monitoring herbicides. Therefore, in this work we have analyzed the molecular reactivity of isoproturon, diuron, linuron, and chlorotoluron employing the global and local reactivity descriptors derived from DFT. However, in aqueous conditions, the molecular basis for the initial degradation of these phenylurea herbicides remains unknown [35, 36]. In this sense, an advantage of the theoretical quantum methods resides in the fact that the electronic properties of the molecules may be calculated with enough accuracy, at the molecular level, to explain adequately the experimental behavior. Thus, it is well known that the Fukui function values obtained from various population schemes may provide negative values; however, the Hirshfeld Stockholder population scheme [70] yields positive values [71]. Thus, nucleophilic and free radical attacks on diuron and linuron would cause a substitution of the hydrogen atom on 7C, while electrophilic attacks on diuron and chlorotoluron would cause the elimination of the dimethylurea fragment.
Dihedral angle 2C-4C-10N-12C = 17.53, 10N-12C-13N-21O = 18.34. Evaluation and characterization of a commercial immunosorbent cartridge for the solid-phase extraction of phenylureas from aqueous matrices. A. EU Annex III PIC DGD) (, R = Peer reviewed scientific publications, L = Pesticide manuals and hard copy reference books / other sources, A = Chromosome aberration (EFSA database), E = Unspecified genotoxicity type (miscellaneous data source), (Also known as: IPU; ipuron; CGA 18731; AE F016410), A herbicide for use, typically, in cereals to control annual grasses and many broad-leaved weeds, Cereals including wheat, barley, rye; Rice-wheat cropping systems, EC Regulation 1107/2009 (repealing 91/414), Approved for use () under EC 1107/2009 in the following EU Member States, International Chemical Identifier key (InChIKey), International Chemical Identifier (InChI), InChI=1S/C12H18N2O/c1-9(2)10-5-7-11(8-6-10)13-12(15)14(3)4/h5-9H,1-4H3,(H,13,15), Cambridge Crystallographic Data Centre diagrams. Selective, systemic absorbed by roots and leaves. 1SD, UK +44 (0)1223 49 44 44, Copyright EMBL-EBI 2018 | EBI is an outstation of the European Molecular Optical whole-cell biosensor using Chlorella vulgaris designed for monitoring herbicides. Therefore, in this work we have analyzed the molecular reactivity of isoproturon, diuron, linuron, and chlorotoluron employing the global and local reactivity descriptors derived from DFT. However, in aqueous conditions, the molecular basis for the initial degradation of these phenylurea herbicides remains unknown [35, 36]. In this sense, an advantage of the theoretical quantum methods resides in the fact that the electronic properties of the molecules may be calculated with enough accuracy, at the molecular level, to explain adequately the experimental behavior. Thus, it is well known that the Fukui function values obtained from various population schemes may provide negative values; however, the Hirshfeld Stockholder population scheme [70] yields positive values [71]. Thus, nucleophilic and free radical attacks on diuron and linuron would cause a substitution of the hydrogen atom on 7C, while electrophilic attacks on diuron and chlorotoluron would cause the elimination of the dimethylurea fragment. 2021 by the U.S. Secretary of Commerce A selective, systemic herbicide used to control annual grasses and broadleaf
Read the winning articles.
The Fukui function values for chlorotoluron are reported in Figure 6; note that the distribution of the more reactive sites for this herbicide are located on 4C, 7C, and 7C atoms for electrophilic, nucleophilic, and free radical attacks, respectively. Follow the links above to find out more about the data Global reactivity descriptors for isoproturon, chlorotoluron, diuron, and linuron at the MP2/6-311++G(2d,2p) level of theory, in the aqueous phase. Bond distances: 2C-4C = 1.40, 2C-5C = 1.39, 3C-6C = 1.40, 3C-7C = 1.39, 4C-7C = 1.40, 5C-6C = 1.40, 6C-20C = 1.50, 3C-19Cl = 1.76, 4C-10N = 1.41, 10N-12C = 1.39, 12C-18O = 1.24, 12C-13N = 1.36, 13N-24C = 1.45, 13N-14C = 1.46, N-H = 1.00, C-H = 1.09 in methyl groups, and C-H = 1.08 in the ring. The Fukui function can be written as [56]where is the electronic density.
Determination of solid-liquid partition coefficients (Kd) for the herbicides isoproturon and trifluralin in five UK agricultural soils. In the present work, we have analyzed the chemical reactivity of isoproturon, chlorotoluron, diuron, and linuron herbicides in the aqueous phase employing global and local reactivity parameters derived from the Density Functional Theory. NIST Standard Reference CopyCopied, PUIYMUZLKQOUOZ-UHFFFAOYSA-N
errors or omissions in the Database. such sites.  However, a study of the pinpoint distribution of the reactivity of these PHUs requires making precise measurements at the molecular level which may become experimentally complicated. structure cas uhfffaoysa registry chemical number cgi Determination of the herbicide isoproturon in cereal grains and pasta by LC with LC/MS confirmation.
However, a study of the pinpoint distribution of the reactivity of these PHUs requires making precise measurements at the molecular level which may become experimentally complicated. structure cas uhfffaoysa registry chemical number cgi Determination of the herbicide isoproturon in cereal grains and pasta by LC with LC/MS confirmation. 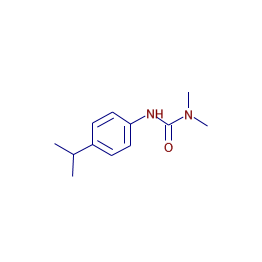
isoproturon This page was last edited on 10 November 2020, at 21:31. The Fukui function, equation (5), can be evaluated by using a finite difference approximation. The results suggest that, in aqueous conditions, chlorotoluron, linuron, and diuron herbicides may be degraded by elimination of urea moiety through electrophilic attacks. reference mzcloud Influence of kinetic sorption and diffusion on pesticide movement through aggregated soils. Technology, Office of Data Luis Humberto Mendoza-Huizar wishes to thank National Laboratory for the Characterization of Physicochemical Properties and Molecular Structure (LACAPFEM) for providing supercomputing time. Data from NIST Standard Reference Database 69: The National Institute of Standards and Technology (NIST) The final atomic charges for isoproturon, chlorotoluron, diuron, and linuron were obtained in the framework of MP2 theory; in all calculations of these atomic charges, the option DENSITY = MP2 was used. Contribution of non-agricultural pesticides to pesticide load in surface water. Chemical Reactivity of Isoproturon, Diuron, Linuron, and Chlorotoluron Herbicides in Aqueous Phase: A Theoretical Quantum Study Employing Global and Local Reactivity Descriptors, Centro de Investigaciones Qumicas, Universidad Autnoma del Estado de Hidalgo, Carretera Pachuca-Tulancingo Km 4.5, 42186 Mineral de la Reforma, HGO, Mexico, http://ec.europa.eu/environment/water/water-framework/priority_substances.htm, http://www.cdc.gov/niosh/docs/81-123/pdfs/0247.pdf, S. Giacomazzi and N. Cochet, Environmental impact of diuron transformation: a review,, S. R. Srensen, C. N. Albers, and J. Aamand, Rapid mineralization of the phenylurea herbicide diuron by, L. Vrielynck, C. Lapouge, S. Marquis, J. Kister, and N. Dupuy, Theoretical and experimental vibrational study of phenylurea: structure, solvent effect and inclusion process with the, F. Javier Benitez, C. Garcia, J. L. Acero, and F. J. ejpau ipu urea ia structure Real, Removal of phenylurea herbicides from waters by using chemical oxidation treatments,, M. Kaur, A. K. Malik, and B. Singh, Determination of phenylurea herbicides in tap water and soft drink samples by HPLC-UV and solid-phase extraction,, J. M. Sanchis-Mallols, S. Sagrado, M. J. Medina-Hernndez, R. M. Villanueva Camaas, and E. Bonet-Domingo, Determination of phenylurea herbicides in drinking waters by HPLC and solid phase extraction,, N. Badawi, S. Rnhede, S. Olsson et al., Metabolites of the phenylurea herbicides chlorotoluron, diuron, isoproturon and linuron produced by the soil fungus, M. Carrier, M. Besson, C. Guillard, and E. Gonze, Removal of herbicide diuron and thermal degradation products under Catalytic Wet Air Oxidation conditions,. reals, its use within the EU has been banned after September 2017 on the grounds of potential groundwater contamination and risks to aquatic life; there have also been concerns about its endocrine-dis
Degradation of pesticides in biobeds: the effect of concentration and pesticide mixtures. analytical This service is an Elixir Core Data Resource.
pesticide Dihedral angle 2C-4C-10N-12C = 40.3.
A lysimeter experiment to investigate temporal changes in the availability of pesticide residues for leaching.


 In Figures 5, 6, 7, and 8 the values of the condensed Fukui function for isoproturon, chlorotoluron, diuron, and linuron are reported, respectively. the 2015, Article ID 751527, 9 pages, 2015. https://doi.org/10.1155/2015/751527, 1Centro de Investigaciones Qumicas, Universidad Autnoma del Estado de Hidalgo, Carretera Pachuca-Tulancingo Km 4.5, 42186 Mineral de la Reforma, HGO, Mexico. III.
In Figures 5, 6, 7, and 8 the values of the condensed Fukui function for isoproturon, chlorotoluron, diuron, and linuron are reported, respectively. the 2015, Article ID 751527, 9 pages, 2015. https://doi.org/10.1155/2015/751527, 1Centro de Investigaciones Qumicas, Universidad Autnoma del Estado de Hidalgo, Carretera Pachuca-Tulancingo Km 4.5, 42186 Mineral de la Reforma, HGO, Mexico. III.  Modeling of an annular photocatalytic reactor for water purification: oxidation of pesticides.
Modeling of an annular photocatalytic reactor for water purification: oxidation of pesticides.