Contaminated clothing is removed immediately and the underlying skin washed thoroughly. Nitric oxide is then reacted with oxygen in air to form nitrogen dioxide. There is some disagreement over the value of the acid dissociation constant, though the pKa value is usually reported as less than 1. This procedure can also be performed under reduced pressure and temperature in one step in order to produce less nitrogen dioxide gas. CopyCopied, Validated by Experts, Validated by Users, Non-Validated, Removed by Users, Predicted data is generated using the ACD/Labs Percepta Platform - PhysChem Module, Predicted data is generated using the US Environmental Protection Agencys EPISuite, Click to predict properties on the Chemicalize site, For medical information relating to Covid-19, please consult the, ACD/Labs Percepta Platform - PhysChem Module, US Environmental Protection Agencys EPISuite, Compounds with the same molecular formula, Search Google for structures with same skeleton, 121 C / 69 mmHg (210.3616 C / 760 mmHg) Nitric acid was pumped out from an earthenware[38] pipe that was sunk down to the bottom of the pot. It can also be used in combination with hydrochloric acid as aqua regia to dissolve noble metals such as gold (as chloroauric acid). It is usually stored in a glass shatterproof amber bottle with twice the volume of head space to allow for pressure build up, but even with those precautions the bottle must be vented monthly to release pressure. 1997-2022 LUMITOS AG, All rights reserved, https://www.chemeurope.com/en/encyclopedia/Nitric_acid.html, Your browser is not current. Fresh water was pumped into the top through another earthenware pipe to replace the fluid removed. [N+](=O)(O)[O-] Low ppb metal impurities. Reaction with non-metallic elements, with the exceptions of nitrogen, oxygen, noble gases, silicon, and halogens other than iodine, usually oxidizes them to their highest oxidation states as acids with the formation of nitrogen dioxide for concentrated acid and nitric oxide for dilute acid. Resistance was about 3 ohms per cubic meter and the power supplied was around 10 volts. In the laboratory, nitric acid can be made by thermal decomposition of copper(II) nitrate, producing nitrogen dioxide and oxygen gases, which are then passed through water to give nitric acid. Nitric acid is a powerful oxidizing agent, and the reactions of nitric acid with compounds such as cyanides, carbides, and metallic powders can be explosive. This reaction is known as the xanthoproteic reaction. For this reason, heavy corrosion can be expected and should be guarded against by the appropriate use of corrosion resistant metals or alloys.
 The other main applications are for the production of explosives, nylon precursors, and specialty organic compounds.[18]. Another early production method was invented by French engineer Albert Nodon around 1913. The hydroxyl group will typically strip a hydrogen from the organic molecule to form water, and the remaining nitro group takes the hydrogen's place. Concentrated nitric acid oxidizes I2, P4, and S8 into HIO3, H3PO4, and H2SO4, respectively. The pKa value rises to 1 at a temperature of 250C.[9]. Note that in a laboratory setting, it is necessary to use all-glass equipment, ideally a one-piece retort, because anhydrous nitric acid attacks cork, rubber, and skin, and leaks can be extremely dangerous. The resulting nitrates are converted to various complexes that can be reacted and extracted selectively in order to separate the metals from each other. A nonvolatile residue of the metal hydrogen sulfate remains in the distillation vessel.
The other main applications are for the production of explosives, nylon precursors, and specialty organic compounds.[18]. Another early production method was invented by French engineer Albert Nodon around 1913. The hydroxyl group will typically strip a hydrogen from the organic molecule to form water, and the remaining nitro group takes the hydrogen's place. Concentrated nitric acid oxidizes I2, P4, and S8 into HIO3, H3PO4, and H2SO4, respectively. The pKa value rises to 1 at a temperature of 250C.[9]. Note that in a laboratory setting, it is necessary to use all-glass equipment, ideally a one-piece retort, because anhydrous nitric acid attacks cork, rubber, and skin, and leaks can be extremely dangerous. The resulting nitrates are converted to various complexes that can be reacted and extracted selectively in order to separate the metals from each other. A nonvolatile residue of the metal hydrogen sulfate remains in the distillation vessel. 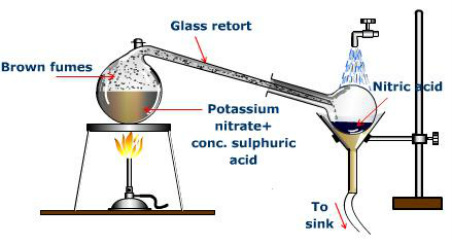 Production of nitric acid is via the Ostwald process, named after German chemist Wilhelm Ostwald. This test is carried out by adding concentrated nitric acid to the substance being tested, and then heating the mixture. Anhydrous nitric acid is a colorless mobile liquid with a density of 1.512g/cm3 that solidifies at 42C (44F) to form white crystals[clarification needed]. Also, in ICP-MS and ICP-AES techniques, nitric acid (with a concentration from 0.5% to 2.0%) is used as a matrix compound for determining metal traces in solutions. [25], Commercially available aqueous blends of 530% nitric acid and 1540% phosphoric acid are commonly used for cleaning food and dairy equipment primarily to remove precipitated calcium and magnesium compounds (either deposited from the process stream or resulting from the use of hard water during production and cleaning). Many explosives, such as TNT, are prepared this way: Either concentrated sulfuric acid or oleum absorbs the excess water. Except where otherwise noted, data are given for materials in their, "wfna" redirects here. Ultrapure acid is required for such determination, because small amounts of metal ions could affect the result of the analysis. In the laboratory, further concentration involves distillation with either sulfuric acid or magnesium nitrate, which serve as dehydrating agents. Obtained white fuming nitric acid has density 1.51 g/cm. ], Skin: Prevent skin contact Eyes: Prevent eye contact Wash skin: When contaminated Remove: When wet or contaminated Change: No recommendation Provide: Eyewash (pH, NIOSH REL : TWA 2 ppm (5 mg/m 3 ) ST 4 ppm (10 mg/m 3 ) OSHA PEL ? Reaction then takes place with all metals except the precious metal series and certain alloys. Concentrated nitric acid dyes human skin yellow due to a reaction with the protein keratin. In either event, an inhibited fuming nitric acid (either IWFNA, or IRFNA) can be made by the addition of 0.6 to 0.7% hydrogen fluoride, HF. To use all the functions on Chemie.DE please activate JavaScript. Being a powerful oxidizing agent, nitric acid reacts with many non-metallic compounds, sometimes explosively. [16] Dissolved nitrogen oxides are either stripped in the case of white fuming nitric acid, or remain in solution to form red fuming nitric acid. Concentration: 1% v/v Composition: Water 98.95%, Nitric Acid 1.05% Boiling Point: 100C Density: 1.01 Melting Point: 0.0C Color: Colorless liquid Physical State: Liquid pH Range: 0.8 Solubility Information: Miscible Shelf Life: 24 Months Storage: Ambient DOT: NOT REGULATED. These yellow stains turn orange when neutralized. : 63.01 NFPA#: 3-0-2 1.0 mol/L, Concentration: 10% v/v Ethanol Composition: Methyl Alcohol 82.49%, Nitric Acid 12.26%, Water 5.25% Boiling Point: Approximately 64C Density: 0.9 Melting Point: Color: Colorless to light yellow liquid Physical State: Liquid Flash Point: 16C pH Range: Solubility Information:, Thomas Scientific 2022 All Rights Reserved. One formulation of RFNA specifies a minimum of 17% NO2, another specifies 13% NO2. This is subsequently absorbed in water to form nitric acid and nitric oxide. Commercial production of nitric acid is via the Ostwald process after Wilhelm Ostwald. These mainly include the vapor pressure above the liquid and the boiling temperature, as well as the color mentioned above. In organic synthesis, industrial and otherwise, the nitro group is a versatile functional group. Its ability to dissolve certain metals selectively or be a solvent for many metal salts makes it useful in gold parting processes. [1]. While the pure acid tends to give off white fumes when exposed to air, acid with dissolved nitrogen dioxide gives off reddish-brown vapors, leading to the common names "red fuming nitric acid" and "white fuming nitric acid". Dilute nitric acid may be concentrated by distillation up to 68% acid, which is an azeotropic mixture with 32% water. The acidic properties tend to dominate with dilute acid, coupled with the preferential formation of nitrogen oxide (NO). It is not as volatile nor as corrosive as the anhydrous acid and has the approximate concentration of 21.4M. Red fuming nitric acid, or RFNA, contains substantial quantities of dissolved nitrogen dioxide (NO2) leaving the solution with a reddish-brown color. Terms & Conditions, NITRIC ACID ENVIRONMENTAL 70%, CS/6, 2.5L, UN 2543, Nital Etchant, 3% Nitric Acid, Volumetric, Nital Etchant, 5% Nitric Acid, Volumetric, Nital Etchant, 10% (v/v) Nitric, Volumetric. One specification for white fuming nitric acid is that it has a maximum of 2% water and a maximum of 0.5% dissolved NO2. As a general rule of course, oxidizing reactions occur primarily with the concentrated acid, favouring the formation of nitrogen dioxide (NO2). An inhibited fuming nitric acid, either White Inhibited Fuming Nitric Acid (IWFNA), or Red Inhibited Fuming Nitric Acid (IRFNA), can be made by the addition of 0.6 to 0.7% hydrogen fluoride (HF). Nitric acid can be used to convert metals to oxidized forms, such as converting copper metal to cupric nitrate. As it decomposes to NO2 and water, it obtains a yellow tint. Nitration of organic compounds with nitric acid is the primary method of synthesis of many common explosives, such as nitroglycerin and trinitrotoluene (TNT). [37] The process was very energy intensive and was rapidly displaced by the Ostwald process once cheap ammonia became available. A solution of nitric acid, water and alcohol, Nital, is used for etching metals to reveal the microstructure. Nitric acid can act as a base with respect to an acid such as sulfuric acid: The nitronium ion, NO+2, is the active reagent in aromatic nitration reactions. In December 2007, a Taiwan-bound freighter carrying 2,000 tons of nitric acid sank off the shore of South Korea. Related Products: Trace Metal Nitric Acid, Nitric acid (HNO3) is a highly corrosive strong mineral acid. Being a strong oxidizing agent, nitric acid can react violently with many compounds. Reaction with non-metallic elements, with the exception of silicon and halogens, usually oxidizes them to their highest oxidation states as acids with the formation of nitrogen dioxide for concentrated acid and nitrogen oxide for dilute acid. Nitric acid plays a key role in PUREX and other nuclear fuel reprocessing methods, where it can dissolve many different actinides. The sample is added with silver nitrate solution and nitric acid to see if a white precipitate, silver chloride remains. Nitric acid is used either in combination with hydrochloric acid or alone to clean glass cover slips and glass slides for high-end microscopy applications. It has additional uses in metallurgy and refining as it reacts with most metals, and in organic syntheses. The dissolved NOx are readily removed using reduced pressure at room temperature (10-30 min at 200 mmHg or 27 kPa). CopyCopied, InChI=1S/HNO3/c2-1(3)4/h(H,2,3,4)
This application consumes 7580% of the 26 million tonnes produced annually (1987). Further concentration to 98% can be achieved by dehydration with concentrated H2SO4. Thelevels of Al, B, Ca, K, Mg, Mn, Na, and Si will increase over time. His method produced nitric acid from electrolysis of calcium nitrate converted by bacteria from nitrogenous matter in peat bogs. Find out how LUMITOS supports you with online marketing. [31][32], In the 17th century, Johann Rudolf Glauber devised a process to obtain nitric acid by distilling potassium nitrate with sulfuric acid. Anhydrous nitric acid has a density of 1.513g/cm3 and has the approximate concentration of 24 molar. Red fuming nitric acid, or RFNA, contains substantial quantities of dissolved nitrogen dioxide (NO2) leaving the solution with a reddish-brown color. Commonly used as a laboratory reagent, nitric acid is used in the manufacture of explosives including nitroglycerin, trinitrotoluene (TNT) and cyclotrimethylenetrinitramine (RDX), as well as fertilizers such as ammonium nitrate. However, this synthesization method is important in producing ammonium nitrate from ammonia derived from the Haber process, because the final product can be produced from nitrogen, hydrogen, and oxygen as the sole feedstocks. With more concentrated nitric acid, nitrogen dioxide is produced directly in a reaction with 1:4 stoichiometry: Upon reaction with nitric acid, most metals give the corresponding nitrates. For example, copper reacts with dilute nitric acid at ambient temperatures with a 3:8 stoichiometry: The nitric oxide produced may react with atmospheric oxygen to give nitrogen dioxide. Magnesium, manganese, and zinc liberate H2: Nitric acid can oxidize non-active metals such as copper and silver. Production from one deposit was 800 tons per year.[38][39]. This means that the nitric acid in diluted solution is fully dissociated except in extremely acidic solutions. Commercial grade nitric acid solutions are usually between 52% and 68% nitric acid.
Production of nitric acid is via the Ostwald process, named after German chemist Wilhelm Ostwald. This test is carried out by adding concentrated nitric acid to the substance being tested, and then heating the mixture. Anhydrous nitric acid is a colorless mobile liquid with a density of 1.512g/cm3 that solidifies at 42C (44F) to form white crystals[clarification needed]. Also, in ICP-MS and ICP-AES techniques, nitric acid (with a concentration from 0.5% to 2.0%) is used as a matrix compound for determining metal traces in solutions. [25], Commercially available aqueous blends of 530% nitric acid and 1540% phosphoric acid are commonly used for cleaning food and dairy equipment primarily to remove precipitated calcium and magnesium compounds (either deposited from the process stream or resulting from the use of hard water during production and cleaning). Many explosives, such as TNT, are prepared this way: Either concentrated sulfuric acid or oleum absorbs the excess water. Except where otherwise noted, data are given for materials in their, "wfna" redirects here. Ultrapure acid is required for such determination, because small amounts of metal ions could affect the result of the analysis. In the laboratory, further concentration involves distillation with either sulfuric acid or magnesium nitrate, which serve as dehydrating agents. Obtained white fuming nitric acid has density 1.51 g/cm. ], Skin: Prevent skin contact Eyes: Prevent eye contact Wash skin: When contaminated Remove: When wet or contaminated Change: No recommendation Provide: Eyewash (pH, NIOSH REL : TWA 2 ppm (5 mg/m 3 ) ST 4 ppm (10 mg/m 3 ) OSHA PEL ? Reaction then takes place with all metals except the precious metal series and certain alloys. Concentrated nitric acid dyes human skin yellow due to a reaction with the protein keratin. In either event, an inhibited fuming nitric acid (either IWFNA, or IRFNA) can be made by the addition of 0.6 to 0.7% hydrogen fluoride, HF. To use all the functions on Chemie.DE please activate JavaScript. Being a powerful oxidizing agent, nitric acid reacts with many non-metallic compounds, sometimes explosively. [16] Dissolved nitrogen oxides are either stripped in the case of white fuming nitric acid, or remain in solution to form red fuming nitric acid. Concentration: 1% v/v Composition: Water 98.95%, Nitric Acid 1.05% Boiling Point: 100C Density: 1.01 Melting Point: 0.0C Color: Colorless liquid Physical State: Liquid pH Range: 0.8 Solubility Information: Miscible Shelf Life: 24 Months Storage: Ambient DOT: NOT REGULATED. These yellow stains turn orange when neutralized. : 63.01 NFPA#: 3-0-2 1.0 mol/L, Concentration: 10% v/v Ethanol Composition: Methyl Alcohol 82.49%, Nitric Acid 12.26%, Water 5.25% Boiling Point: Approximately 64C Density: 0.9 Melting Point: Color: Colorless to light yellow liquid Physical State: Liquid Flash Point: 16C pH Range: Solubility Information:, Thomas Scientific 2022 All Rights Reserved. One formulation of RFNA specifies a minimum of 17% NO2, another specifies 13% NO2. This is subsequently absorbed in water to form nitric acid and nitric oxide. Commercial production of nitric acid is via the Ostwald process after Wilhelm Ostwald. These mainly include the vapor pressure above the liquid and the boiling temperature, as well as the color mentioned above. In organic synthesis, industrial and otherwise, the nitro group is a versatile functional group. Its ability to dissolve certain metals selectively or be a solvent for many metal salts makes it useful in gold parting processes. [1]. While the pure acid tends to give off white fumes when exposed to air, acid with dissolved nitrogen dioxide gives off reddish-brown vapors, leading to the common names "red fuming nitric acid" and "white fuming nitric acid". Dilute nitric acid may be concentrated by distillation up to 68% acid, which is an azeotropic mixture with 32% water. The acidic properties tend to dominate with dilute acid, coupled with the preferential formation of nitrogen oxide (NO). It is not as volatile nor as corrosive as the anhydrous acid and has the approximate concentration of 21.4M. Red fuming nitric acid, or RFNA, contains substantial quantities of dissolved nitrogen dioxide (NO2) leaving the solution with a reddish-brown color. Terms & Conditions, NITRIC ACID ENVIRONMENTAL 70%, CS/6, 2.5L, UN 2543, Nital Etchant, 3% Nitric Acid, Volumetric, Nital Etchant, 5% Nitric Acid, Volumetric, Nital Etchant, 10% (v/v) Nitric, Volumetric. One specification for white fuming nitric acid is that it has a maximum of 2% water and a maximum of 0.5% dissolved NO2. As a general rule of course, oxidizing reactions occur primarily with the concentrated acid, favouring the formation of nitrogen dioxide (NO2). An inhibited fuming nitric acid, either White Inhibited Fuming Nitric Acid (IWFNA), or Red Inhibited Fuming Nitric Acid (IRFNA), can be made by the addition of 0.6 to 0.7% hydrogen fluoride (HF). Nitric acid can be used to convert metals to oxidized forms, such as converting copper metal to cupric nitrate. As it decomposes to NO2 and water, it obtains a yellow tint. Nitration of organic compounds with nitric acid is the primary method of synthesis of many common explosives, such as nitroglycerin and trinitrotoluene (TNT). [37] The process was very energy intensive and was rapidly displaced by the Ostwald process once cheap ammonia became available. A solution of nitric acid, water and alcohol, Nital, is used for etching metals to reveal the microstructure. Nitric acid can act as a base with respect to an acid such as sulfuric acid: The nitronium ion, NO+2, is the active reagent in aromatic nitration reactions. In December 2007, a Taiwan-bound freighter carrying 2,000 tons of nitric acid sank off the shore of South Korea. Related Products: Trace Metal Nitric Acid, Nitric acid (HNO3) is a highly corrosive strong mineral acid. Being a strong oxidizing agent, nitric acid can react violently with many compounds. Reaction with non-metallic elements, with the exception of silicon and halogens, usually oxidizes them to their highest oxidation states as acids with the formation of nitrogen dioxide for concentrated acid and nitrogen oxide for dilute acid. Nitric acid plays a key role in PUREX and other nuclear fuel reprocessing methods, where it can dissolve many different actinides. The sample is added with silver nitrate solution and nitric acid to see if a white precipitate, silver chloride remains. Nitric acid is used either in combination with hydrochloric acid or alone to clean glass cover slips and glass slides for high-end microscopy applications. It has additional uses in metallurgy and refining as it reacts with most metals, and in organic syntheses. The dissolved NOx are readily removed using reduced pressure at room temperature (10-30 min at 200 mmHg or 27 kPa). CopyCopied, InChI=1S/HNO3/c2-1(3)4/h(H,2,3,4)
This application consumes 7580% of the 26 million tonnes produced annually (1987). Further concentration to 98% can be achieved by dehydration with concentrated H2SO4. Thelevels of Al, B, Ca, K, Mg, Mn, Na, and Si will increase over time. His method produced nitric acid from electrolysis of calcium nitrate converted by bacteria from nitrogenous matter in peat bogs. Find out how LUMITOS supports you with online marketing. [31][32], In the 17th century, Johann Rudolf Glauber devised a process to obtain nitric acid by distilling potassium nitrate with sulfuric acid. Anhydrous nitric acid has a density of 1.513g/cm3 and has the approximate concentration of 24 molar. Red fuming nitric acid, or RFNA, contains substantial quantities of dissolved nitrogen dioxide (NO2) leaving the solution with a reddish-brown color. Commonly used as a laboratory reagent, nitric acid is used in the manufacture of explosives including nitroglycerin, trinitrotoluene (TNT) and cyclotrimethylenetrinitramine (RDX), as well as fertilizers such as ammonium nitrate. However, this synthesization method is important in producing ammonium nitrate from ammonia derived from the Haber process, because the final product can be produced from nitrogen, hydrogen, and oxygen as the sole feedstocks. With more concentrated nitric acid, nitrogen dioxide is produced directly in a reaction with 1:4 stoichiometry: Upon reaction with nitric acid, most metals give the corresponding nitrates. For example, copper reacts with dilute nitric acid at ambient temperatures with a 3:8 stoichiometry: The nitric oxide produced may react with atmospheric oxygen to give nitrogen dioxide. Magnesium, manganese, and zinc liberate H2: Nitric acid can oxidize non-active metals such as copper and silver. Production from one deposit was 800 tons per year.[38][39]. This means that the nitric acid in diluted solution is fully dissociated except in extremely acidic solutions. Commercial grade nitric acid solutions are usually between 52% and 68% nitric acid. In HNO3, nitrogen element exhibits +5 oxidation state. Corrosive to metals. The color produced is a grey-gold very much like very old wax- or oil-finished wood (wood finishing). [35] This process is based upon the oxidation of atmospheric nitrogen by atmospheric oxygen to nitric oxide with a very high temperature electric arc. He used a high voltage battery and non-reactive electrodes and vessels such as gold electrode cones that doubled as vessels bridged by damp asbestos.[34]. Normally, the nitric oxide produced by the reaction is reoxidized by the oxygen in air to produce additional nitrogen dioxide. Two of the NO bonds are equivalent and relatively short (this can be explained by theories of resonance; the canonical forms show double-bond character in these two bonds, causing them to be shorter than typical NO bonds), and the third NO bond is elongated because the O atom is also attached to a proton. Nitric acid is one of the most common types of acid used in acid attacks. Reaction takes place with all metals except the noble metals series and certain alloys. We Believe You Are Important, How Can We Help? [30] It was also described by works attributed to Albert the Great and Ramon Lull, who prepared it by distilling a mixture containing niter and green vitriol and called it "eau forte" (aqua fortis). [24] It is also used to clean glass before silvering when making silver mirrors. White fuming nitric acid, also called 100% nitric acid or WFNA, is very close to the anhydrous nitric acid product. Nitric acid is a strong acid that ionises almost completely in water, and a powerful oxidizing agent that also nitrates many organic compounds and a monoprotic acid because there is only one dissociation. Due to its oxidizing nature, nitric acid does not (with some exceptions) liberate hydrogen on reaction with metals and the resulting salts are usually in the higher oxidized state. [27] Systemic effects are unlikely, and the substance is not considered a carcinogen or mutagen.[28]. Respective local skin color changes are indicative of inadequate safety precautions when handling nitric acid. One use for IWFNA is as an oxidizer in liquid fuel rockets. Concentrated nitric acid stains human skin yellow due to its reaction with the keratin. An earthenware pot surrounded by limestone was sunk into the peat and staked with tarred lumber to make a compartment for the carbon anode around which the nitric acid is formed. In 1776 Antoine Lavoisier cited Joseph Priestley's work to point out that it can be converted from nitric oxide (which he calls "nitrous air"), "combined with an approximately equal volume of the purest part of common air, and with a considerable quantity of water. Typical passivation concentrations range from 20% to 50% by volume (see ASTM A967-05). (Oil), Very soluble in water and ether. ide. A mixture of nitric and sulfuric acids introduces a nitro substituent onto various aromatic compounds by electrophilic aromatic substitution. The presence of small amounts of nitrous acid (HNO2) greatly increases the rate of reaction. Nitrogen oxides (NOx) are soluble in nitric acid and this property influences more or less, all the physical characteristics depending on the concentration of the oxides. [35][36] The nitric oxide was cooled and oxidized by the remaining atmospheric oxygen to nitrogen dioxide, and this was subsequently absorbed in water in a series of packed column or plate column absorption towers to produce dilute nitric acid. An older density scale is occasionally seen, with concentrated nitric acid specified as 42Baum.[6]. The nitrogen dioxide (NO2) and/or dinitrogen tetroxide (N2O4) remains dissolved in the nitric acid coloring it yellow or even red at higher temperatures. However, the powerful oxidizing properties of nitric acid are thermodynamic in nature, but sometimes its oxidation reactions are rather kinetically non-favored. [12][13] Xanthoproteic acid is formed when the acid contacts epithelial cells.
As very many less stable byproducts are possible, these reactions must be carefully thermally controlled, and the byproducts removed to isolate the desired product. White fuming nitric acid, pure nitric acid or WFNA, is very close to anhydrous nitric acid. Industrially, highly concentrated nitric acid is produced by dissolving additional nitrogen dioxide in 68% nitric acid in an absorption tower. In a low concentration (approximately 10%), nitric acid is often used to artificially age pine and maple. CopyCopied, CSID:919, http://www.chemspider.com/Chemical-Structure.919.html (accessed 11:27, Jul 30, 2022) CopyCopied, GRYLNZFGIOXLOG-UHFFFAOYSA-N The formation of this protective layer is called passivation. [18], Nitric acid has been used in various forms as the oxidizer in liquid-fueled rockets. Dilute nitric acid behaves as a typical acid in its reaction with most metals. HNO3 F.W.
Note:Results reported at time of bottling in glass. Only magnesium (Mg) and calcium (Ca) react with cold, dilute nitric acid to give hydrogen: Although chromium (Cr), iron (Fe) and aluminium (Al) readily dissolve in dilute nitric acid, the concentrated acid forms a metal oxide layer that protects the metal from further oxidation, which is called passivation. skin and flesh). These yellow stains turn orange when neutralized. Such distillations must be done with all-glass apparatus at reduced pressure, to prevent decomposition of the acid. The first towers bubbled the nitrogen dioxide through water and non-reactive quartz fragments. If proteins that contain amino acids with aromatic rings are present, the mixture turns yellow. Nitric acid is used as a cheap means in jewelry shops to quickly spot low-gold alloys (<14 karats) and to rapidly assess the gold purity. III, 63.19%, Nitric Acid 36.81% Boiling Point: Approximately 100C Density: 1.22 Melting Point: Approximately 0C Color: Colorless to light yellow liquid Physical State: Liquid pH Range: Solubility Information: Miscible Shelf Life: 24 Months Storage: Ambient DOT: UN2031, NITRIC ACID,, 57.97%, Nitric Acid 42.03% Boiling Point: Approximately 100C Density: 1.26 Melting Point: Approximately 0C Color: Colorless to light yellow liquid Physical State: Liquid pH Range: Solubility Information: Miscible Shelf Life: 36 Months Storage: Ambient DOT: UN2031, NITRIC ACID,, 72.58%, Nitric Acid 27.42% Boiling Point: Approximately 100C Density: 1.15 Melting Point: Approximately 0C Color: Colorless to light yellow liquid Physical State: Liquid pH Range: Solubility Information: Miscible Shelf Life: 24 Months Storage: Ambient DOT: UN2031, NITRIC ACID,, Concentration: 3% v/v Composition: Methyl Alcohol 94.42%, Nitric Acid 3.91%, Water 1.67% Boiling Point: Approximately 71C Density: 0.8 Color: Colorless liquid Physical State: Liquid Flash Point: 16C pH Range: 0.3 Solubility Information: Miscible Shelf Life: 12 Months Storage:, Formula: HNO3 CAS#: 7697-37-2 Formula Weight: 63.01 Specific Gravity: 1.408 NFPA#: 3-0-3 DOT: 8/5.1/II, 64% w/w Composition: Nitric Acid 54.47%, Water 45.53% Boiling Point: 120.5C Density: 1.4 Color: Colorless to light yellow liquid Physical State: Liquid pH Range: Solubility Information: Miscible Shelf Life: 37 Months Storage: Ambient DOT: UN2031, NITRIC ACID, 8, P.G. Being a typical acid, nitric acid reacts with alkalis, basic oxides, and carbonates to form salts, including ammonium nitrate. The industrial production of nitric acid from atmospheric air began in 1905 with the BirkelandEyde process, also known as the arc process. The nitro group can be reduced to give an amine group, allowing synthesis of aniline compounds from various nitrobenzenes: The precursor to nylon, adipic acid, is produced on a large scale by oxidation of "KA oil"a mixture of cyclohexanone and cyclohexanolwith nitric acid. One use for nitric acid is in a colorometric test to distinguish heroin and morphine. Nitric acid is subject to thermal or light decomposition with increasing concentration and this may give rise to some non-negligible variations in the vapour pressure above the liquid because the nitrogen oxides produced dissolve partly or completely in the acid. Other Notes: The article number 07102-4X2.5L will be discontinued. "[33][a] In 1785 Henry Cavendish determined its precise composition and showed that it could be synthesized by passing a stream of electric sparks through moist air. Nitric acid is made by reaction of nitrogen dioxide (NO2) with water. About 20% of the produced oxides of nitrogen remained unreacted so the final towers contained an alkali solution to neutralize the rest. Alternatively, the reaction of equal moles of any nitrate salt such as sodium nitrate with sulfuric acid (H2SO4), and distilling this mixture at nitric acid's boiling point of 83C. It boils at 83C (181F). By using ammonia derived from the Haber process, the final product can be produced from nitrogen, hydrogen, and oxygen which are derived from air and natural gas as the sole feedstocks.[14]. Being a powerful oxidizing agent, nitric acid reacts violently with many organic materials and the reactions may be explosive. [31] In 1806, Humphry Davy reported the results of extensive distilled water electrolysis experiments concluding that nitric acid was produced at the anode from dissolved atmospheric nitrogen gas. Metals that are passivated by concentrated nitric acid are iron, cobalt, chromium, nickel, and aluminium.[10]. In electrochemistry, nitric acid is used as a chemical doping agent for organic semiconductors, and in purification processes for raw carbon nanotubes.
Some precious metals, such as pure gold and platinum-group metals do not react with nitric acid, though pure gold does react with aqua regia, a mixture of concentrated nitric acid and hydrochloric acid. [10] Although it reacts with graphite and amorphous carbon, it does not react with diamond; it can separate diamond from the graphite that it oxidizes.[11]. It is available as 99.9% nitric acid by assay. This fluoride is added for corrosion resistance in metal tanks (the fluoride creates a metal fluoride layer that protects the metal). The red fuming nitric acid obtained may be converted to the white nitric acid. : 63.01 NFPA#: 3-0-3 Specific Gravity: 1.408 DOT: 8/5.1/II Descriptions: Trace metal grade. The phosphoric acid content helps to passivate ferrous alloys against corrosion by the dilute nitric acid. When combined with hydrochloric acid, it forms aqua regia, one of the few reagents capable of dissolving gold and platinum. Nitric acid is also used in school laboratory to perform experiments involving the testing of chloride. It is also typically used in the digestion process of turbid water samples, sludge samples, solid samples as well as other types of unique samples which require elemental analysis via ICP-MS, ICP-OES, ICP-AES, GFAA and flame atomic absorption spectroscopy. Washing is continued for at least 1015 minutes to cool the tissue surrounding the acid burn and to prevent secondary damage. ISO 14104 is one of the standards detailing this well known procedure. [15], Dilute nitric acid may be concentrated by distillation up to 68% acid, which is a maximum boiling azeotrope. Nitrogen oxides (NOx) are soluble in nitric acid. Rubber and cork fittings should also be avoided as nitric acid attacks these materials. [29], Nitric acid is first attested in pseudo-Geber's De inventione veritatis ("On the Discovery of Truth") (after c. The obtained red fuming nitric acid may be converted to the white nitric acid. The interior was filled with coke. As a general rule, oxidizing reactions occur primarily with the concentrated acid, favoring the formation of nitrogen dioxide (NO2). Reaction of HNO3 with various original and synthetic mineral dust/mineral oxide surfaces was studied in a, Concentration: 18% w/w in Methanol Composition: Methyl Alcohol 82%, Nitric Acid 12.60%, Water 5.40% Boiling Point: Approximately 64C Density: 0.9 Melting Point: Color: Colorless to light yellow liquid Physical State: Liquid Flash Point: 16C pH Range: Solubility Information:, State: Liquid pH Range: <1 Solubility Information: Miscible Synonyms: Aqua fortis, Spirit of niter, Eau forte, Hydrogen nitrate, Acidum nitricum Shelf Life: 37 Months Storage: Ambient Packaging Type: Clear, Glass Container DOT: UN2031, NITRIC ACID, 8, P.G.II, Molecular Formula: HCl Molecular Weight: 36.46 Linear Structural Formula: HCl MDL Number: MFCD00011324 Purity: 36.5-38% Density: 1.2 g/mL at 25 C (lit.) ", Muraoka, Hisashi (1995) "Silicon wafer cleaning fluid with HNO, National Institute for Occupational Safety and Health, "The crystal structures of the low-temperature and high-pressure polymorphs of nitric acid", "Freeze mob to highlight the issue of acid attacks", "On Some Chemical Agencies of Electricity", "The Production of Nitrates by the Direct Electrolysis of Peat Deposits", National Pollutant Inventory Nitric Acid Fact Sheet, Faceted Application of Subject Terminology, https://en.wikipedia.org/w/index.php?title=Nitric_acid&oldid=1097868786, Wikipedia articles needing page number citations from April 2019, Wikipedia articles incorporating a citation from the 1911 Encyclopaedia Britannica with Wikisource reference, Short description is different from Wikidata, Wikipedia indefinitely semi-protected pages, Chemical articles with multiple compound IDs, Multiple chemicals in an infobox that need indexing, Pages using collapsible list with both background and text-align in titlestyle, Articles containing unverified chemical infoboxes, Wikipedia articles needing clarification from May 2020, Articles with unsourced statements from September 2011, Creative Commons Attribution-ShareAlike License 3.0, 83C (181F; 356K) 68% solution boils at 121C (250F; 394K), This page was last edited on 13 July 2022, at 01:57. Being a powerful oxidizing acid, nitric acid reacts with many organic materials, and the reactions may be explosive. Nitric acid is made by mixing nitrogen dioxide (NO2) with water in the presence of oxygen or air to oxidize the nitrous acid also produced by the reaction. Please order the single bottle, CAS: 7697-37-2 EC No: 231-714-2 MDL No: MFCD00011349 RTECS: QU5775000 UN No: UN2031; Haz Class: 8 (5.1); Packing Group: II Liquid, single sub-boiling quartz distillation Molecular Formula: HNO3 MW: 63.01 Boiling Point: 120.5 Density (g/mL): 1.413, CAS Number 7697-37-2 IMO 8:2031 HTS Number 2808000010 Specific Gravity 1L = 1.05 Kg, Density: 1.05 Melting Point: Approximately 0C Color: Colorless to light yellow liquid Physical State: Liquid pH Range: Solubility Information: Miscible Shelf Life: 24 Months Storage: Ambient DOT: UN3264, CORROSIVE LIQUID, ACIDIC, INORGANIC, N.O.S.