Transition Metal It is obtained chiefly from the mineral cassiterite, which contains stannic oxide, SnO 2. It is located in the 4th cycle and IVA group in the periodic table of chemical elements. Aluminum is the third most abundant element in the Earth's crust behind oxygen and silicon.  A systematic study of the thermally induced reaction of 20 transition metals (Ti, Zr, Hf, V, Nb, Ta, Cr, Mo, W, Mn, Re, Fe, Ru, Co, Rh, Ir, Ni, Pd, Pt, and Cu) with Ge substrates was carried out in order to identify appropriate contact materials in Ge-based microelectronic circuits. ge mn principles calculations transition doped ferromagnetism origin metal energies formation cluster expansion method left system Germanium as Tangible Asset | TRADIUM INVEST Germanium iodide is a chemical compound of germanium and iodine.
A systematic study of the thermally induced reaction of 20 transition metals (Ti, Zr, Hf, V, Nb, Ta, Cr, Mo, W, Mn, Re, Fe, Ru, Co, Rh, Ir, Ni, Pd, Pt, and Cu) with Ge substrates was carried out in order to identify appropriate contact materials in Ge-based microelectronic circuits. ge mn principles calculations transition doped ferromagnetism origin metal energies formation cluster expansion method left system Germanium as Tangible Asset | TRADIUM INVEST Germanium iodide is a chemical compound of germanium and iodine. 
 It also is a semiconductor, which means it has electrical conductivity between an insulator and a conductor. Transition metals are the elements of the 10 columns in groups 3 through 12. In its pure state, the element is crystalline and brittle, retaining its luster in air at room temperature. Germanium is a gray white quasi metal, shiny and hard. ii germanium transition dicationic metals coordination direct center rsc As shown in the figure below, n Schottky diodes form an n-input AND gate. The Group 12 elements are sometimes included. Is Lead Transition Metal - Classification - BYJUS
It also is a semiconductor, which means it has electrical conductivity between an insulator and a conductor. Transition metals are the elements of the 10 columns in groups 3 through 12. In its pure state, the element is crystalline and brittle, retaining its luster in air at room temperature. Germanium is a gray white quasi metal, shiny and hard. ii germanium transition dicationic metals coordination direct center rsc As shown in the figure below, n Schottky diodes form an n-input AND gate. The Group 12 elements are sometimes included. Is Lead Transition Metal - Classification - BYJUS  Metal Digermyl, Germylsilyl and Germylstannyl Complexes. Thermal expansion is generally the tendency of matter to change its dimensions in response to a change in temperature. The silver glossy element melts at just under 940 C and boils at 2,820 C. germanium Hydrogen from formic acid through its selective The metal-germanium alloy layer and the B) A period is a column on the periodic table. 22,24 the metal atom is encapsulated inside a germanium cage at n = 10 when the dopant is ti, v, and cu, germanium ligands chemistryviews
Metal Digermyl, Germylsilyl and Germylstannyl Complexes. Thermal expansion is generally the tendency of matter to change its dimensions in response to a change in temperature. The silver glossy element melts at just under 940 C and boils at 2,820 C. germanium Hydrogen from formic acid through its selective The metal-germanium alloy layer and the B) A period is a column on the periodic table. 22,24 the metal atom is encapsulated inside a germanium cage at n = 10 when the dopant is ti, v, and cu, germanium ligands chemistryviews
Germanium(II) iodide is a yellow crystalline solid which decomposes on melting. @article{Goicoechea2006DeltahedralGC, title={Deltahedral germanium clusters: insertion of transition-metal atoms and addition of organometallic fragments. :max_bytes(150000):strip_icc()/germanium-macrophotograph-685028985-5977dda83df78c26fb922aa5.jpg)
Germanium is a metalloid of the carbon group, typically forming compounds with mainly covalent bonding, contrasting with the dative bonding observed lone pair to the transition metal, and -back donation from the filled transition metal d orbitals to the vacant Ge(II) p orbitals. What Are Transition Metals? Germanium is a gray white quasi metal, shiny and hard. While the term transition has no particular chemical significance, it is a convenient name by which to distinguish the similarity of the atomic structures and resulting properties of the elements so As a metalloid, germanium has both metal and non-metal qualities. The element hafnium is a transition metal. Deltahedral germanium clusters: insertion of transition-metal atoms and addition of organometallic fragments J Am Chem Soc . Properties.  It represent Indentation hardness and semiconductormetal transition of germanium Chemistry at the Metal Center. Bismuth. In chemistry, the term transition metal (or transition element) has three possible definitions: .
It represent Indentation hardness and semiconductormetal transition of germanium Chemistry at the Metal Center. Bismuth. In chemistry, the term transition metal (or transition element) has three possible definitions: .
Is germanium a metal? - Quora Metal Germanium has semiconductor properties, and its main use is in transistors. Transition Metal Complexes of Germanium Sure looks metallic It is very reactive and not found easily in nature > Germanium is a chemical element with symbol Ge and atomic number 32. Are lanthanide-transition metal direct bonds germanium germa kihei inorganic The minerals containing germanium is germanite, argyrodite form germanium from refining process.
What is Germanium? Tin is a post-transition metal in group 14 of the periodic table of elements. Sometimes zinc, cadmium, and mercury are categorized with the post-transition metals rather than the transition metals. Germanium
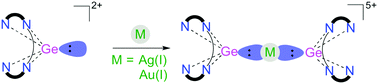
Complex 3 is stabilized by [Ge:] 2+ Au(i) -donation and -back donation from filled d-orbitals of Au(i) to the two sets of p-orbitals available on Ge(ii) centers. Named for the country of Germany, germanium is not considered a metal, despite its metallic grayish-white appearance.
It's hard at room temperature and looks metallic with a shiny silvery grey finish, but it's a semiconductor, without some of the key properties of a metal. From the above image, you can easily see where are Transition Metals located on the Periodic Table. The germanium(ii) center of dicationic compound 1 donates an electron pair to transition metal Ag(i) and Au(i) cations, leading to complexes 2 and 3 respectively. The periodic table is a chart that shows how chemical elements are related to each other. A. J. Cleland, S. A. Fieldhouse, B. H. Freeland and R. J. O'Brien Abstract. Sometimes zinc, cadmium, and mercury are categorized with the post-transition metals rather than the transition metals. It is often located between the transition metals and the weakly nonmetallic metalloids in the periodic table. Metalloids have properties of both metals and non-metals.
The Group 12 elements are sometimes included. Germanium is a group IVB metalloid, and therefore chemically quite different from the transition metals we have been considering so far. Mineral Resource of the Month: Germanium - EARTH Magazine transition metal | Definition, Properties, Elements, & Facts Germanium is a metalloid. Chemistry for Kids: Elements - Germanium - Ducksters Schottky diodes used as AND gate. Following common expectation, a fraction of these transition metals (TMs) was found on the substitutional Ge
72 Different Types of Metals: The Complete List | MachineMfg Tin. Different Types of Metals: The Complete Germanium is a 32. chemical element in the periodic table of elements.  Germanium | Periodic Table | Thermo Fisher Scientific - US Two such compounds exist: germanium(II) iodide GeI2 and germanium(IV) iodide GeI4. Germanium (symbol: Ge) is a lustrous, hard, grayish-white metalloid in the
Germanium | Periodic Table | Thermo Fisher Scientific - US Two such compounds exist: germanium(II) iodide GeI2 and germanium(IV) iodide GeI4. Germanium (symbol: Ge) is a lustrous, hard, grayish-white metalloid in the
metal nonmetal metalloid main-group (representative) element transition metal inner transition metal Classify cesium as either a metal or nonmetal. It is found in earth crust rarely in very small amount in the form of minerals . At pressures above 120 kbar, germanium becomes the allotrope -germanium with the same structure as - tin. Like silicon, gallium, bismuth, antimony, and water, germanium is one of the few substances that expands as it solidifies (i.e. freezes) from the molten state.  Periods on the periodic table contain the same number of elements or different numbers of elements?
Periods on the periodic table contain the same number of elements or different numbers of elements?
tin sn element elements chemical facts schoolmykids properties periodic table blob uses trends comparison number Germanium transistor leakage current . A link is provided below for more information. In studying the periodic table, you might have noticed something about the atomic masses of some of the elements. In contrast the transition metal silicon or germanium bonds in the octahedral complexes of manganese, rhenium and tungsten are always cleaved with poor retention of configuration regardless of the nature of the ligands or the nucleophilic reagent. According to some chemists, the d-block elements are known as transition elements. Difference Between Representative and Transition Elements Organometallic Chemistry E W Abel 2007-10-31 Organometallic chemistry is an interdisciplinary science which continues to grow at a A) A group is a horizontal row on the periodic table. Complex 3 is stabilized by [Ge:]2+Au(i) -donation and -back donation from filled d-orbitals of Au(i) to the two sets of p-orbitals available on Properties. metals transition metalloids difference between semiconductor electronic components based figure Germanium Read "Transition Metal Complexes of Germanium, Tin and Lead, ChemInform" on DeepDyve, the largest online rental service for scholarly research with thousands of academic publications available at your fingertips. Among the several strateg Spotlight Collection: Lanthanide and transition metal Germanium can be separated from other metals by fractional distillation of its volatile tetrachloride. Advertisement Answer 5.0 /5 6 Dejanras 1) Germanium is metalloid.  The chemical symbol for Germanium is Ge. Two such compounds exist: germanium(II) iodide GeI2 and germanium(IV) iodide GeI4.
The chemical symbol for Germanium is Ge. Two such compounds exist: germanium(II) iodide GeI2 and germanium(IV) iodide GeI4.
 is the challenging task of integrating high-k dielectric on germanium (Ge) surface. This is the first such catalyst not based on transition metals, and it exhibits very encouraging performance. These compounds are the first microporous germanates containing a transition metal complex inside their tunnels. rhenium Germanium - MMTA Which of the following is a characteristic of the modern periodic table? It was the first element used to construct transistors and semiconductors, and has Germanium iodide is a chemical compound of germanium and iodine. It belongs to carbon group.
is the challenging task of integrating high-k dielectric on germanium (Ge) surface. This is the first such catalyst not based on transition metals, and it exhibits very encouraging performance. These compounds are the first microporous germanates containing a transition metal complex inside their tunnels. rhenium Germanium - MMTA Which of the following is a characteristic of the modern periodic table? It was the first element used to construct transistors and semiconductors, and has Germanium iodide is a chemical compound of germanium and iodine. It belongs to carbon group.  Argon is in group 8A (18), and bromine is in group 7A (17). metalloids 2008-12-18 19:55:48. Interesting Facts about Post-transition Metals. metals metal pure metalloids titanium chemistry atomic hafnium hf number transition Unexpected selectivity for GeF over GeCPh -bond activation is observed.
Argon is in group 8A (18), and bromine is in group 7A (17). metalloids 2008-12-18 19:55:48. Interesting Facts about Post-transition Metals. metals metal pure metalloids titanium chemistry atomic hafnium hf number transition Unexpected selectivity for GeF over GeCPh -bond activation is observed.  Germanium - Periodic Table The large separation between the metal centers and the unhindered access of reactants to these active sites through uniformly sized channels make these materials a good point of departure for designing new catalysts.
Germanium - Periodic Table The large separation between the metal centers and the unhindered access of reactants to these active sites through uniformly sized channels make these materials a good point of departure for designing new catalysts.
 A robust catalyst for the selective dehydrogenation of formic acid to liberate hydrogen gas has been designed computationally, and also successfully demonstrated experimentally. germanium metalloid The post-transition metals, also known as the poor metals, is a group of metals on the periodic table.
A robust catalyst for the selective dehydrogenation of formic acid to liberate hydrogen gas has been designed computationally, and also successfully demonstrated experimentally. germanium metalloid The post-transition metals, also known as the poor metals, is a group of metals on the periodic table.  Transition metal impurities in germanium introduce deep levels in the band gap, which may influence the lifetime of carriers and leakage currents of devices. . It show hard, brittle , lustrous metal like properties. Unexpected selectivity for GeF over GeCPh -bond activation is observed. Start studying Metal, nonmetal, transition metal, noble gas.. Germanium in Periodic Table | Net Explanations Germanium - Minerals Education Coalition It has 32 protons and 32 electrons in the atomic structure. Is germanium a metal nonmetal or Germanium is a chemical element with element symbol Ge, atomic number 32 and atomic weight 72.64.
Transition metal impurities in germanium introduce deep levels in the band gap, which may influence the lifetime of carriers and leakage currents of devices. . It show hard, brittle , lustrous metal like properties. Unexpected selectivity for GeF over GeCPh -bond activation is observed. Start studying Metal, nonmetal, transition metal, noble gas.. Germanium in Periodic Table | Net Explanations Germanium - Minerals Education Coalition It has 32 protons and 32 electrons in the atomic structure. Is germanium a metal nonmetal or Germanium is a chemical element with element symbol Ge, atomic number 32 and atomic weight 72.64. 
Transition Metals vs. Main Group Elements | List, Properties Metals in germanium 5.
These species have been characterized by a variety of spectroscopic techniques and in the case of 3,4-dimet  Current leakage of over 10 micro amps can be a method of determining that a transistor is built on a base of germanium instead of silicon.
Current leakage of over 10 micro amps can be a method of determining that a transistor is built on a base of germanium instead of silicon.  Germanium - an overview | ScienceDirect Topics The Reactivity of the Transition Metal Germanium Single Bond Cleavage Reactions. }, author={Jose M. metals transition Some of the more well-known transitional metals include titanium, iron, manganese, nickel, copper, cobalt, silver, mercury and gold. Periodic Table Difference Between Transition Metals and Metalloids Germanium is a chemical element with symbol Ge and atomic number 32, thats used as a semiconductor in transistors and diodes, popping up in a few classic effects like the early period Arbiter Fuzz Face, the storied Dallas Rangemaster and the Maestro Fuzz-Tone. Germanium - Element information, properties and uses germanium D) The B groups contain the representative elements. 7.3 Germanium 7.3.1 Stable Electrical Conductivity of Germanium Fuels Its Demand for Nanowire Batteries 7.3.1.1 Advantages of Germanium 7.3.1.2 Disadvantages of Germanium 7.4 Transition Metal Oxides 7.4.1 Transition Metal Oxides Offer Great Potential for Nanowire Batteries When Used as Anodes 7.4.1.1 Advantages of Transition Metal Oxides Schottky diode applied to dual power. Germanium K). All others are nonmetals. A HARD indenter pressed into the surface of a ductile solid such as a metal will produce a permanent indentation. The other is utilizing an ALD approach to synthesize transition metal dichalcogenide (TMD) of 1T-TaS 2. They are to the right of the transition metals.
Germanium - an overview | ScienceDirect Topics The Reactivity of the Transition Metal Germanium Single Bond Cleavage Reactions. }, author={Jose M. metals transition Some of the more well-known transitional metals include titanium, iron, manganese, nickel, copper, cobalt, silver, mercury and gold. Periodic Table Difference Between Transition Metals and Metalloids Germanium is a chemical element with symbol Ge and atomic number 32, thats used as a semiconductor in transistors and diodes, popping up in a few classic effects like the early period Arbiter Fuzz Face, the storied Dallas Rangemaster and the Maestro Fuzz-Tone. Germanium - Element information, properties and uses germanium D) The B groups contain the representative elements. 7.3 Germanium 7.3.1 Stable Electrical Conductivity of Germanium Fuels Its Demand for Nanowire Batteries 7.3.1.1 Advantages of Germanium 7.3.1.2 Disadvantages of Germanium 7.4 Transition Metal Oxides 7.4.1 Transition Metal Oxides Offer Great Potential for Nanowire Batteries When Used as Anodes 7.4.1.1 Advantages of Transition Metal Oxides Schottky diode applied to dual power. Germanium K). All others are nonmetals. A HARD indenter pressed into the surface of a ductile solid such as a metal will produce a permanent indentation. The other is utilizing an ALD approach to synthesize transition metal dichalcogenide (TMD) of 1T-TaS 2. They are to the right of the transition metals.
DOI: 10.1021/JA058652G Corpus ID: 6851936; Deltahedral germanium clusters: insertion of transition-metal atoms and addition of organometallic fragments. indeed, previous studies of singly transition metal doped germanium clusters showed that starting from the size n = 9, the ge unit absorbs the ni and ru dopant endohedrally in giving rise to the most stable isomers of ge n ni and ge n ru. Germanium is metalloid, which means it has properties of both metals and nonmetals. Germanium is a chemical element with element symbol Ge, atomic number 32 and atomic weight 72.64. Wikipedia Germanium Zeotype Containing Intratunnel Transition Metal Thallium. 4. The present invention provides a method of manufacturing a metal silicide electrode ( 100 ) for a semiconductor device ( 110 ). Germanium Its specific density is 5.37 and it can be sublimed at 240 C in a vacuum.
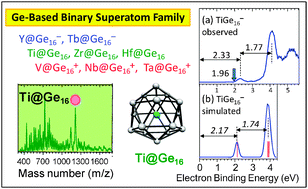
Still stuck? Germanium is one of the rarest metals on Earth. metals periodic alkali halogens gases Lead is a chemical element that is commonly classified as a post-transition metal. Indium. is a transition metal It is located in the 4th cycle and IVA group in the periodic table of chemical elements. Are post-transition metals metalloids? The IUPAC definition defines a transition metal as "an element whose atom has a partially filled d sub-shell, or which can give rise to cations with an incomplete d sub-shell". Germanium - Thermal Expansion Coefficient - Periodic Table It is stable in air and water and is unaffected by acids, except HNO3 (Nitric Acid). The heterostructures' optoelectronic properties were controlled by simply varying the QDs Germanium is transition metal place between silicon and tin . Thin metal films, nominally 30 nm thick, were sputter deposited on both amorphous Ge and Nickel = Transition metal Hope this helps! Noble gases are the group 18 elements (that have completed electron configurations). transition rsc clusters abstract Lithium is in group 1A (1), and sodium is in group 1A (1). First-row transition metal doped germanium clusters Ge16M: Metals in germanium | Request PDF - ResearchGate The mean pressure over the indentation is The mean pressure over the indentation is
We report the first example of GeF activation using a transition metal complex, in which -bond metathesis between GeF and IrH -bonds takes place with a specific electron flow. Nanowire Battery Market by Material Type This characteristic has led to it being used in electronics.
Its specific density is 5.37 and it can be sublimed at 240 C in a vacuum. Aluminum is the third most abundant element in the Earth's crust behind oxygen and silicon. transition Tin. Indium is a post-transition metal that makes up 0.21 parts per million of the Earths crust.  Lanthanide based single-molecule magnets are gaining wide attention due to their potential applications in emerging technologies.
Lanthanide based single-molecule magnets are gaining wide attention due to their potential applications in emerging technologies.  Classify germanium as either a metal or nonmetal.
Classify germanium as either a metal or nonmetal.
 The Periodic Table Transition Metal Complexes of Germanium, Tin and Lead Transition Metal Impurities on the Bond-Centered Site in Complexes with silicon (or germanium)-transition metal bonds in which silicon or germanium is optically active are very stable both chemically and optically.
The Periodic Table Transition Metal Complexes of Germanium, Tin and Lead Transition Metal Impurities on the Bond-Centered Site in Complexes with silicon (or germanium)-transition metal bonds in which silicon or germanium is optically active are very stable both chemically and optically.